"un métabolon pour la synthèse d'ubiquinone" publié dans Cell Chemical Biology
"un métabolon pour la synthèse d'ubiquinone" publié dans Cell Chemical Biology

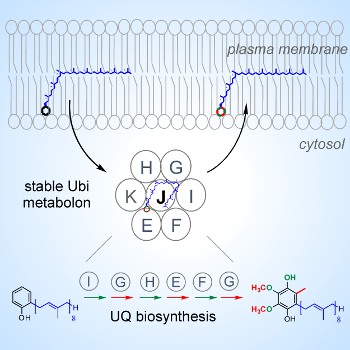
Ubiquinone is a conserved isoprenoid lipid of crucial physiological importance. Isoprenoid intermediates in ubiquinone biosynthesis are extremely hydrophobic; thus the pathway is proposed to be membrane-associated. Unexpectedly, we demonstrate that most steps in bacterial UQ biosynthesis are independent of the membrane and occur inside a soluble multiprotein complex that forms a stable metabolon. We elucidate a key role for the SCP2 domain of the UbiJ protein in binding the isoprenoid intermediates inside the multiprotein complex, documenting that the widespread SCP2 domain also functions in assisting chemical modifications of lipids. Our work highlights a remarkable bacterial metabolon synthesizing the hydrophobic ubiquinone molecule in the cytosol, and raises questions regarding the trafficking of isoprenoid lipids in and out of the membrane.
Cell Chem Biol. 2019, 26(4):482-492